
Group photo. From left to right: Montserrat Argandoña, Joaquín J. Nieto, Francine Piubeli, Carmen Vargas, Rosa García, Emilia Naranjo, Alí Tahrioui and Manuel Salvador.
Our research group (Group BIO-320 of the Ministry of Economy, Innovation, Science and Employment of the Junta de Andalucía) aims to study the molecular genetics of osmoadaptation processes in prokaryotes, specifically in a group of microorganisms Extremophiles, moderate halophilic bacteria, and their application in the production of useful biostabilizing compounds in the Pharmaceutical, Agriculture, and Biomedical industries. I know It is a Group, led by Profs. Carmen Vargas Macías and Joaquín J. Nieto, made up of 7 doctors and two graduates, who have extensive experience in this line of research, subsidized in the last 20 years by numerous national and international projects of the National Plan, the Andalusian Government and of the European Commission.
Chromohalobacter salexigens, is a gamma-proteobacterium that is considered an excellent biological model to study osmoadaptation processes in bacteria, since it has one of the widest known growth salt ranges (0.5 to 3 M NaCl in minimal medium). ). In order to compensate for this osmotic stress, it is capable of intracellularly accumulating large amounts of compatible solutes, low molecular weight polar compounds, either after transport from the external medium if they are present, as is the case with betaine, or through their synthesis. , in the absence of said compounds in the medium. This strategy is very versatile, since it allows it to adapt to very different external salinities by adjusting the intracellular concentration of said compatible solutes, since it can also expel them to the outside if the osmotic pressure decreases in the external environment. The compounds that it synthesizes mainly in response to osmotic and thermal stress are the ectoines (ectoine and hydroxyectoine), some diamino acids that have acquired great industrial interest in recent years. Indeed, these compounds, which can only be obtained by biological synthesis, have excellent biostabilizing and protective properties, which is why they are used in Dermopharmacy as components of anti-aging gels and creams, protection creams against UV rays, etc., and in Molecular Biology as protectors of antibodies, enzymes, nucleic acids, among others, also presenting enormous potential in the field of Biomedicine. Thus, it has recently been described that they inhibit in vitro the formation of betaamyloid aggregates characteristic of neurodegenerative diseases such as Alzheimer's. Since ectoines accumulate mainly at high salinity and temperature, C. salexigens it is an excellent example of a cellular factory for the production of the same. Among the advantages it presents is the ease of cultivation using conventional media, its great metabolic versatility, the availability of genetic tools for its manipulation (most of them developed by our Group) and, very importantly, the fact that the sequence is available. complete of its genome.
In recent years, we have carried out numerous studies aimed at elucidating the molecular mechanisms of osmoadaptation in C. salexigens. Thus, we have shown that, in addition to certain modifications in the lipid composition of its membranes, this bacterium is also capable of synthesizing trehalose involved in protection against high temperature and desiccation, through genes otsAB, as well as betaine, from its precursor, choline, through the genes betIBA. We have also characterized at the molecular level the genes for the synthesis of ectoin (ectABC and its hydroxy derivative, hydroxyectoin (ectD, ectE), as well as the genes that encode the uptake systems of the same from the external environment (teaABC). The synthesis of ectoines in C. salexigens As well as its regulation, it is extraordinarily complex, being osmo- and thermoregulated, and in which different regulators intervene (sS, s32, Fur, etc.), having demonstrated for the first time in halophilic bacteria the interrelation between iron homeostasis and osmoadaptation
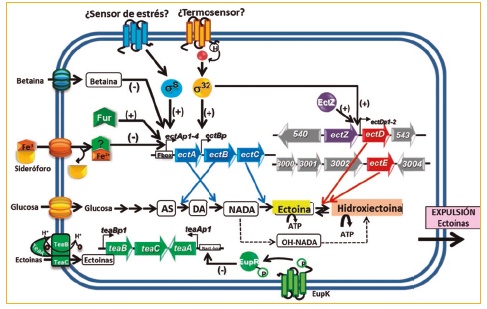
Figure 1. Diagram of the osmoadaptation mechanisms of C. salexigens
We have recently characterized at the molecular level a response regulator (EupR) of a two-component system involved in osmodetection, the first described for halophilic bacteria, which seems to mediate the transcriptional control of the ectoin uptake system, encoded by genes. teaABC. In addition, there are alternative synthesis and degradation routes, currently under study (Figure 1). All this makes it very difficult to obtain mutant strains that produce ectoines that are capable of superproducing these compounds under more favorable conditions at an industrial level than the current ones, such as, for example, at room temperature or in low salinity conditions. , our Group has addressed this objective in recent years through the Systems Biology approach, in which data from the application of last frontier Molecular Biology techniques, such as proteomics, transcriptomics, metabolomics and flowomics, with bioinformatic modeling methods in silico (Fig. 2).
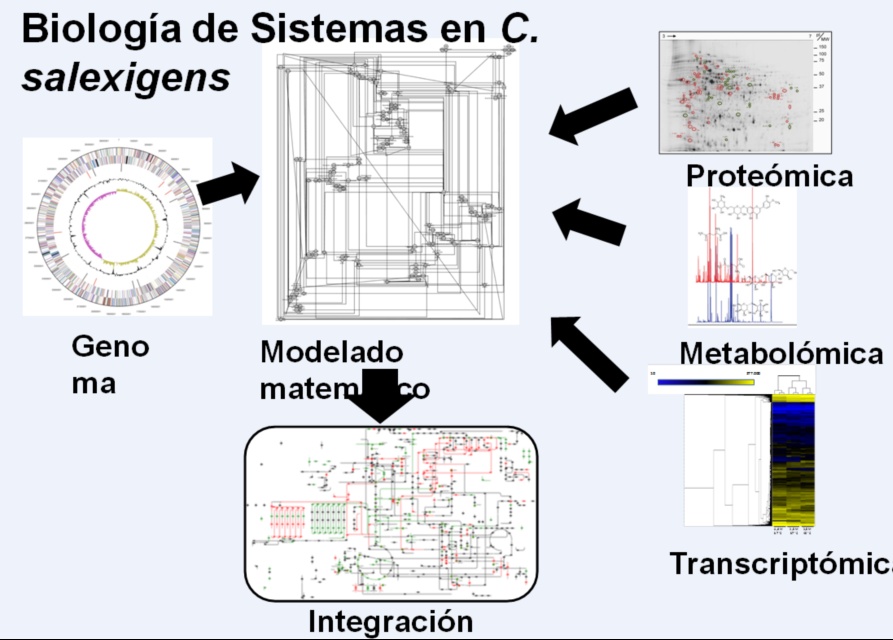
Figure 2. Systems Biology of C. salexigens
In this regard, we have completed the first genomic-scale refined metabolic reconstruction of C. salexigens, in which the metabolism of C and N has been connected with all the routes related to the transport, synthesis, degradation and efflux of the main compatible solutes accumulated by this bacterium.This metabolic network has been used to obtain a first mathematical metabolic model that reflects the biological system of study with enough precision, since it has been manually revised based on previous biochemical and physiological studies, and data from multimic analysis recently carried out by our Group. It has also been validated by comparing the results of the simulations in silico with data from experimental results (determination of growth kinetics with different sources of carbon and energy and nitrogen at different salinities, analysis of extracellular metabolites, determination of enzymatic activities of central metabolism, differential proteomics, among others).
All the information obtained from this model will help us to better predict the physiological responses and thus develop new strategies to optimize the obtaining of strains of C. salexigens hyperproducers of ectoines.
On the other hand, we have obtained new hydroxyectoine-producing mutant strains in which it has been possible to decouple the synthesis from its regulation by temperature and salinity, obtaining production values of this compatible solute at 37ºC and 0.75 M NaCl similar to those of the wild strain at 45º C and 2.5 M NaCl, which is a great advantage from the industrial point of view (results that have been protected under patent). Finally, for the reasons stated above, we are very interested in the molecular study of the neuroprotective effect of ectoines and their possible application in Biomedicine, for which the Group already has collaboration at the level of subsidized research projects with various companies in the sector interested in the subject.
Pastor JM, Bernal V, Salvador M, Argandoña M, Vargas C, Csonka L, Nieto JJ and Cánovas M. (2013). Role ofcentral metabolism in the osmoadaptation of the bacterium C.salexigens. J Biol Chem 288:17769-81.
Rodriguez-Moya J, Argandoña M, Grandson JJ, Churches-War F and Vargas C. (2013). Temperature- and salinity-decoupled overproduction of hydroxyectoine by C. salexigens. Appl Environ Microbiol 79: 1018-1023.
Queen-Good M, Argandoña M, Savior M, Csonka L, Grandson JJ and Vargas C. (2012). Role of trehalose intolerance to salinity, temperature and desiccation in C. salexigens. PLoS One 7(3). 2012e33587.
Argandoña M, Vargas C, Reina-Bueno M, Salvador M and Nieto JJ. (2012). An extended suite of genetic tools for use in bacteria of the Halomonadaceae: an overview. In: P Balbas and A Lorence (eds), Methods in Molecular Biology, Reviews and Protocols, 3rd ed, vol. 824, Humana Press, Totowa, NJ, pp. 167-201.
Argandona M. Nieto JJ, Calderón MI, García-Estepa R and Vargas C. (2011). Interplay between iron homeostasis and the osmostress response in the halophilic C. salexigens. Appl Environ Microbiol 76: 3575-3589.
Copeland A, O’Connor K, et al. (2011). Complete genome sequence of the halophilic and highly halotolerant Chromohalobacter salexigens type strain (1H11T). Stand Genom Sci 5:379-88.
Pastor JM, Salvador M, Argandoña M, Bernal V, Reina-Bueno M, Vargas C, Nieto JJ and Cánovas M. (2010). Ectoines in cell stress protection: uses and biotechnological production. Biotechnol Adv 28: 782-807.
Rodríguez-Moya J, Argandoña M, Reina-Bueno M, Nieto JJ, and Vargas C. (2010). Involvement of a two-component system in the control of the uptake of ectoines by the halophilic bacterium C. salexigens. BMC Microbiol 10: 256-274.
Vargas C, Argandoña M, Garcia-Stepa R, Queen-Good M, Rodriguez-Moya J and Nieto JJ. (2008). Unravelling the adaptation responses to osmotic and temperature stress in C. salexigens, a bacterium with broad salinity tolerance. Saline Systems 4: 14–23.
García-Estepa R, Argandoña M, Reina-Bueno M, Iglesias-Guerra F, Grandson JJ and Vargas C. (2006). The ectD gene, which is involved in the synthesis of the compatible solute hydroxyectoine, is essential for thermoprotection of the halophilic bacterium Chromohalobacter salexigens. J Bacteriol 188: 3774 – 84.
