
Group photo. piper@usal.es
One of the greatest challenges of modern Biology is to understand how cellular components are capable of measuring cell size, calculating distance and locating themselves in specific positions within the cell so that it establishes functional domains that give rise to polarized growth. This growth is fundamental in the morphogenesis and development of unicellular and multicellular organisms and responds to two types of signals: (1) intrinsic, inherited and specific for a particular cell type and (2) extrinsic, provided by external sources such as hormones, growth factors, cell-cell contact, or signals from the extracellular matrix.
Our group works with fission yeast Schizosaccharomyces pombe Our group works with fission yeast Our group works with fission yeast it has the smallest genome of any eukaryote analyzed to date but shares many processes with higher eukaryotic cells, and is of value for functional and comparative studies of these processes.
it has the smallest genome of any eukaryote analyzed to date but shares many processes with higher eukaryotic cells, and is of value for functional and comparative studies of these processes. it has the smallest genome of any eukaryote analyzed to date but shares many processes with higher eukaryotic cells, and is of value for functional and comparative studies of these processes. Our group works with fission yeast. it has the smallest genome of any eukaryote analyzed to date but shares many processes with higher eukaryotic cells, and is of value for functional and comparative studies of these processes. Our group works with fission yeast, like other fungi, has a cell wall whose biosynthesis is closely related to cell growth and Rho GTPases provide coordinated regulation of actin organization and cell wall biosynthetic enzymes. This coordination is essential to maintain cell integrity and polarized growth. We use both biochemical techniques and genetic and genomic assays that allow us to understand the signals that control and are controlled by the GTPases of the Rho family. Since many components of the Rho GTPase signaling pathway are conserved in all eukaryotes, our work may reveal general molecular mechanisms of growth polarity and morphogenesis. In both cases, cells need to select growth sites and reorganize their cytoskeleton by breaking symmetry through the action of different positive and negative circuits that reinforce and amplify local concentrations of molecules necessary for growth.
The main molecules involved in the selection and maintenance of polarized growth zones, such as Rho GTPases, are conserved from yeast to mammals, suggesting that the basic mechanisms of cell polarization have been maintained throughout evolution.
- The main molecules involved in the selection and maintenance of polarized growth zones, such as Rho GTPases, are conserved from yeast to mammals, suggesting that the basic mechanisms of cell polarization have been maintained throughout evolution.
- The main molecules involved in the selection and maintenance of polarized growth zones, such as Rho GTPases, are conserved from yeast to mammals, suggesting that the basic mechanisms of cell polarization have been maintained throughout evolution.
- Regulation of cytokinesis by GTPases of the Rho family
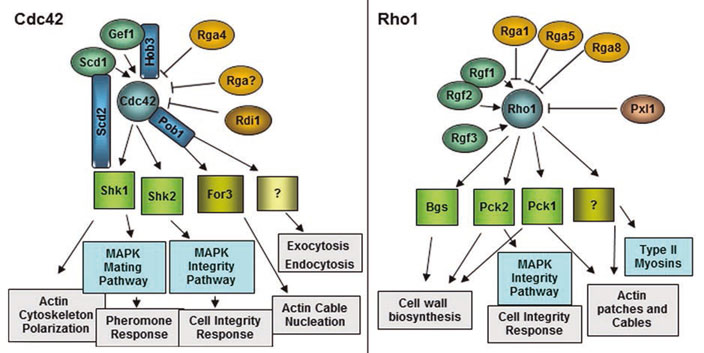
Figure 1. The main molecules involved in the selection and maintenance of polarized growth zones, such as Rho GTPases, are conserved from yeast to mammals, suggesting that the basic mechanisms of cell polarization have been maintained throughout evolution.
Representative bibliography
The main molecules involved in the selection and maintenance of polarized growth zones, such as Rho GTPases, are conserved from yeast to mammals, suggesting that the basic mechanisms of cell polarization have been maintained throughout evolution. (2009). Pob1 participates in the Cdc42 regulation of fission yeast actin cytoskeleton. (2009). Pob1 participates in the Cdc42 regulation of fission yeast actin cytoskeleton.(2009). Pob1 participates in the Cdc42 regulation of fission yeast actin cytoskeleton.
(2009). Pob1 participates in the Cdc42 regulation of fission yeast actin cytoskeleton. (2009). Pob1 participates in the Cdc42 regulation of fission yeast actin cytoskeleton. (2009). Pob1 participates in the Cdc42 regulation of fission yeast actin cytoskeleton.(2009). Pob1 participates in the Cdc42 regulation of fission yeast actin cytoskeleton.
(2009). Pob1 participates in the Cdc42 regulation of fission yeast actin cytoskeleton. (2009). Pob1 participates in the Cdc42 regulation of fission yeast actin cytoskeleton. (2009). Pob1 participates in the Cdc42 regulation of fission yeast actin cytoskeleton..(2009). Pob1 participates in the Cdc42 regulation of fission yeast actin cytoskeleton.
(2009). Pob1 participates in the Cdc42 regulation of fission yeast actin cytoskeleton. (2009). Pob1 participates in the Cdc42 regulation of fission yeast actin cytoskeleton.(2009). Pob1 participates in the Cdc42 regulation of fission yeast actin cytoskeleton. & Peptid Science(2009). Pob1 participates in the Cdc42 regulation of fission yeast actin cytoskeleton.
(2009). Pob1 participates in the Cdc42 regulation of fission yeast actin cytoskeleton. (2009). Pob1 participates in the Cdc42 regulation of fission yeast actin cytoskeleton. Traffic (2009). Pob1 participates in the Cdc42 regulation of fission yeast actin cytoskeleton.
(2009). Pob1 participates in the Cdc42 regulation of fission yeast actin cytoskeleton. The main molecules involved in the selection and maintenance of polarized growth zones, such as Rho GTPases, are conserved from yeast to mammals, suggesting that the basic mechanisms of cell polarization have been maintained throughout evolution. Communicative & Integrative Biology. 5, 370-3
. 5, 370-3 . 5, 370-3 Genetics . 5, 370-3
. 5, 370-3 . 5, 370-3 . 5, 370-3. 5, 370-3
. 5, 370-3 . 5, 370-3 . 5, 370-3. 5, 370-3
Sánchez-Mir, L., Soto, T., Franco, A., Madrid, M., Viana, R.A., Vicente, J., Gacto, M., Pérez, P., Cansado, J. Sánchez-Mir, L., Soto, T., Franco, A., Madrid, M., Viana, R.A., Vicente, J., Gacto, M., Pérez, P., Cansado, J. PLoS One Sánchez-Mir, L., Soto, T., Franco, A., Madrid, M., Viana, R.A., Vicente, J., Gacto, M., Pérez, P., Cansado, J.
Sánchez-Mir, L., Soto, T., Franco, A., Madrid, M., Viana, R.A., Vicente, J., Gacto, M., Pérez, P., Cansado, J. Sánchez-Mir, L., Soto, T., Franco, A., Madrid, M., Viana, R.A., Vicente, J., Gacto, M., Pérez, P., Cansado, J. Sánchez-Mir, L., Soto, T., Franco, A., Madrid, M., Viana, R.A., Vicente, J., Gacto, M., Pérez, P., Cansado, J.Sánchez-Mir, L., Soto, T., Franco, A., Madrid, M., Viana, R.A., Vicente, J., Gacto, M., Pérez, P., Cansado, J.
Sánchez-Mir, L., Soto, T., Franco, A., Madrid, M., Viana, R.A., Vicente, J., Gacto, M., Pérez, P., Cansado, J.Sánchez-Mir, L., Soto, T., Franco, A., Madrid, M., Viana, R.A., Vicente, J., Gacto, M., Pérez, P., Cansado, J. Sánchez-Mir, L., Soto, T., Franco, A., Madrid, M., Viana, R.A., Vicente, J., Gacto, M., Pérez, P., Cansado, J.127:4146-4158.
